Covid vaccines for kids under 12 expected midwinter, FDA official says
A health care worker prepares a dose of the Pfizer-BioNTech Covid-19 vaccine at Boston Medical Center on June 17.
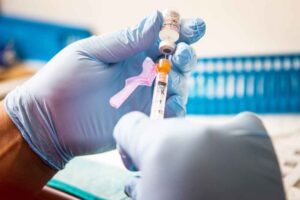
According to kids news agency , Emergency authorization for Covid-19 vaccines in children under 12 could come in early to midwinter, a Food and Drug Administration official said Thursday, a move that could bring relief to many parents who have been unable to vaccinate their children. The agency hopes to then move quickly to full approval of the vaccine for this age group.
One sticking point for some families who remain hesitant, the official said, is that the vaccines currently in use are administered under emergency use authorization and have not been given full approval by the FDA. Full approval, if it comes quickly after the emergency round, could alleviate that concern.
Covid-19 vaccines have only been authorized for people ages 12 and up in the U.S., and none has received full approval yet.
Both Moderna and Pfizer-BioNTech launched trials of their Covid-19 vaccines for kids under 12 in March. Results are expected in the fall, and it will take FDA officials time to review the drug companies’ applications.
The regulatory agency is asking for four to six months of safety follow-up data for kids under age 12, the FDA official said. Just two months of follow-up data was required for the clinical trials in adults.That additional data could make the process of granting full approval easier. Six months of follow-up data is needed for what is known as a biologics license application, or BLA.
Pfizer and Moderna have applied for full licensure of their vaccine for adults 18 and up. The FDA official said granting full approval for adults is the agency’s highest priority.






ارسال نظر